FDA Key Impacts for Food Manufacturing EHS & Operations Teams 2026


FDA's "2026 Food Safety Deliverables"
What EHS and Operations Leaders Must Prepare For Now
The U.S. Food and Drug Administration’s (FDA) Human Foods Program (HFP) 2026 Priority Deliverables document represents one of the most significant shifts in food safety regulation in recent years. For EHS and operations managers in food and beverage manufacturing, these updates are not abstract policy—they signal very real operational, compliance, and risk management changes that will directly impact facilities, processes, and supplier relationships.
The FDA regulates roughly 80% of the U.S. food supply, making its priorities a leading indicator of enforcement trends and compliance expectations.
This article breaks down what matters most—and what actions your facility should take now.
The Big Picture: FDA’s 3 Core Focus Areas for 2026
The 2026 deliverables are organized around three central pillars:
1. Food Chemical Safety
2. Nutrition & Transparency
3. Microbiological Food Safety
These pillars align with the FDA’s broader mission to prevent foodborne illness, reduce chronic disease, and ensure the safety of food ingredients.
For EHS and operations leaders, this translates into increased scrutiny across ingredients, processes, labeling, and facility controls.

1. Chemical Safety: A Major Compliance Shift
GRAS Reform Will Change Ingredient Approval
One of the most disruptive changes is the FDA’s plan to require mandatory submission of GRAS (Generally Recognized as Safe) notices for food ingredients.
Historically, companies could self-determine GRAS status without notifying the FDA. That flexibility is now under pressure.
What this means operationally:
Increased documentation and validation requirements for ingredients
Greater supplier transparency and audit expectations
Potential reformulation of products using legacy or borderline additives
This is not a minor regulatory tweak—it represents a structural change in how ingredients enter and remain in the food supply.
Increased Scrutiny of Food Additives and Chemicals
The FDA is also prioritizing post-market review of food chemicals, including substances already in circulation.
Specific focus areas include:
Synthetic additives and preservatives
Controversial compounds (e.g., phthalates, BHA, BHT)
Petroleum-based food dyes (with plans for removal or replacement)
EHS implications:
Hazard assessments for stored chemicals and ingredients
Review of SDS documentation and chemical inventories
Re-evaluation of storage and segregation practices
Facilities that handle bulk additives or hazardous food chemicals will need to align storage safety with evolving regulatory risk profiles.
MORE FOOD & BEVERAGE SAFETY READING:
Chemical Fire Risk in Ammonia Refrigeration: Lessons from the Koch Foods Fairfield Explosione
2. Nutrition & Labeling: Operational and Brand Impact
Front of Package Labeling is Coming
The FDA plans to implement front-of-package (FOP) nutrition labeling, making it easier for consumers to identify healthier options.
Why this matters beyond marketing:
- 1- Labeling changes can require production line updates
2- Reformulation may be needed to meet “healthier” criteria
3- Increased pressure from retailers and consumers
Focus on Ultra-Processed Foods
The agency is also addressing ultra-processed foods (UPFs) due to their association with chronic disease.
Expected developments include:
1- Potential formal definitions of UPFs
2- Increased regulatory attention and research
3- Future labeling or reformulation pressures
Operational impact:
1- Ingredient sourcing strategies may need adjustment
2- Process complexity could become a compliance factor
3- Transparency in formulations will become more critical
Infant Formula & Nutritional Standards
The FDA is conducting a comprehensive review of infant formula nutrient requirements—the first in decades.
For manufacturers in this category, expect:
1- Stricter formulation standards
2- Increased testing and validation
3- Higher regulatory oversight
3. Microbiological Safety: Expanded Oversight and Inspections
Increased Facility Inspections
The FDA plans to expand inspections by leveraging state resources, effectively increasing oversight across more facilities.
What this means:
1- Higher likelihood of inspections
2- Greater need for audit readiness
3- Expanded documentation requirements
Staying Ahead of the Curve
EHS considerations:
1- Environmental monitoring programs must be robust
2- Cross-contamination controls will face closer scrutiny
3- Airflow, sanitation, and storage practices will be evaluated more aggressively in the future
The FDA is investing in:
1- Improved recall processes – Digital traceability systems
2- Enhanced traceability systems – Supplier tracking
3- Greater transparency in enforcement actions – Rapid response protocols
In Summary
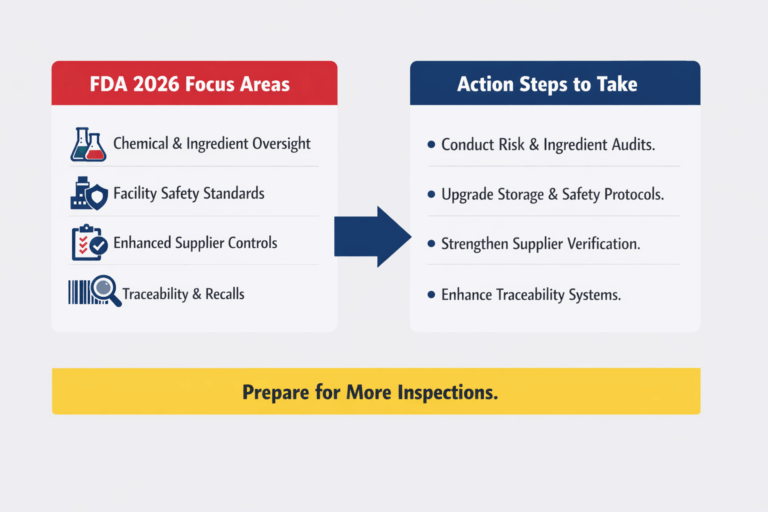
To stay ahead of FDA enforcement trends in 2026, food and beverage manufacturing facilities should take a proactive, structured approach to compliance and risk management. This begins with conducting a thorough chemical and ingredient risk audit, including a detailed review of all additives, preservatives, and processing chemicals to identify any substances that may face increased regulatory scrutiny. Facilities should also evaluate their storage and safety infrastructure to ensure proper containment of hazardous and flammable materials, while confirming alignment with fire codes and ventilation requirements.
In parallel, strengthening supplier verification programs will be critical. This includes requiring comprehensive safety and compliance documentation from vendors and preparing for anticipated changes to GRAS requirements. Upgrading traceability systems is another key priority, with an emphasis on implementing digital tracking solutions that enable rapid and accurate recall capabilities. Finally, organizations should prepare for an increase in FDA inspections by conducting internal audits, standardizing inspection protocols, and training staff to meet evolving compliance expectations.
If your facility isn’t fully aligned with evolving FDA expectations, now is the time to act. Evaluate your current storage, safety, and compliance strategies to identify gaps before they become liabilities. Need guidance on safely managing hazardous chemicals or upgrading your facility for stricter regulations? Connect with our team to explore compliant, scalable solutions designed specifically for food and beverage manufacturing environments. 1-800-233-1480, or fill out the form at right to get your concerns addressed quickly.
References:
FDA Human Foods Program 2026 Priority Deliverables: https://www.fda.gov/about-fda/human-foods-program/human-foods-program-2026-priority-deliverables
Food Processing – “FDA Reveals its 2026 Food Safety To-Do List”: https://www.foodprocessing.com/food-safety/regulatory-compliance/article/55364963/fda-reveals-its-2026-food-safety-to-do-list
